  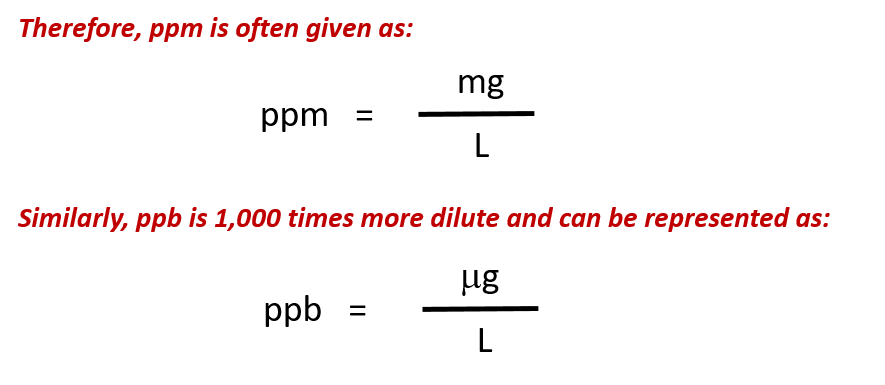
Thus for the 1 M NaCl, the total ion concentration is 2 M for the 1 M CaCl 2, the total ion concentration is 3 M. This definition is equivalent to the following general formula, which is used to calculate ppm concentration from measurements of pollutant mass in a sample. In addition, the total ion concentration is the sum of the individual ion concentrations. .jpg)
However, if the solution were 1 M CaCl 2, there are two Cl −(aq) ions for every formula unit dissolved, so the concentration of Cl −(aq) would be 2 M, not 1 M. For example, if 1 M NaCl were prepared, the solution could also be described as a solution of 1 M Na +(aq) and 1 M Cl −(aq) because there is one Na + ion and one Cl − ion per formula unit of the salt. Notice that the units shall be either gram/mL or kg/L. Because the ions in ionic compounds go their own way when a compound is dissolved in a solution, the resulting concentration of the ion may be different from the concentration of the complete salt. This module is to compute the mass-volume percentage of solute and parts per million (ppm) or to calculate solute or solvent by knowing concentration. What mass of Fe 3+ ion is present in 3,450 mL of H 2O, which has a density of 1.00 g/mL?įor ionic solutions, we need to differentiate between the concentration of the salt versus the concentration of each individual ion. The concentration of Fe 3+ ion in a sample of H 2O is 335.0 ppm. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
